- Genomics
- Transcriptomics
- Epigenomics
- Meta-omics
- Proteomics
- Single-Cell Sequencing
- Immune Repertoire Sequencing
- FFPE Samples

Proteome Profiling
Cases
Technical Information
Contact Us / Wish List

Proteome profiling studies aim to characterize as many proteins as possible from a complete organism, body fluids, or extracts. Profiling results can serve as a foundation for future study of quantitative changes or modifications of target proteins. In combination with transcriptomics analysis, profiling results provide insight into the dynamic changes occurring in a system, allowing for an unbiased view of the state of the system.
Benefits:
- High resolution and accuracy: Based on mass spectrometry with mass accuracy better than 1 ppm and resolving power no less than 100,000 FWHM, the validity of protein identification could be improved in complicated samples through lowering false positive rates.
- High throughput: Thousands of proteins can be identified with a high degree of automation compared to 2D-PAGE.
- Wide application: Theoretically, samples from any species can undergo proteome profiling analysis.
In-depth Proteome Analysis of The Rubber Particle of Hevea Brasiliensis (Para Rubber Tree). Plant Mol Biol. 82(1-2):155-68 (2013).
 The rubber particle is a special organelle in which natural rubber is synthesized and stored in the laticifers of Hevea brasiliensis. To better understand the biological functions of rubber particles and to identify the candidate rubber biosynthesis-related proteins, a comprehensive proteome analysis was performed on H. brasiliensis rubber particles using shotgun, tandem mass-spectrometry profiling approaches. A total of 186 rubber particle proteins were identified, with a range in relative molecular mass of 3.9 to 194.2 kDa and in isoelectric point values of 4.0-11.2. The rubber particle proteins were analyzed for gene ontology and could be categorized into eight major groups according to their functions: including rubber biosynthesis, stress- or defense-related responses, protein processing and folding, signal transduction and cellular transport. These comprehensive proteome data of rubber particles can facilitate investigation of molecular mechanisms of biogenesis, self-homeostasis, and rubber biosynthesis of the rubber particle, and might serve as valuable biomarkers in molecular breeding studies of H. brasiliensis and other alternative rubber-producing species.
The rubber particle is a special organelle in which natural rubber is synthesized and stored in the laticifers of Hevea brasiliensis. To better understand the biological functions of rubber particles and to identify the candidate rubber biosynthesis-related proteins, a comprehensive proteome analysis was performed on H. brasiliensis rubber particles using shotgun, tandem mass-spectrometry profiling approaches. A total of 186 rubber particle proteins were identified, with a range in relative molecular mass of 3.9 to 194.2 kDa and in isoelectric point values of 4.0-11.2. The rubber particle proteins were analyzed for gene ontology and could be categorized into eight major groups according to their functions: including rubber biosynthesis, stress- or defense-related responses, protein processing and folding, signal transduction and cellular transport. These comprehensive proteome data of rubber particles can facilitate investigation of molecular mechanisms of biogenesis, self-homeostasis, and rubber biosynthesis of the rubber particle, and might serve as valuable biomarkers in molecular breeding studies of H. brasiliensis and other alternative rubber-producing species.
The Oyster Genome Reveals Stress Adaptation and Complexity of Shell Formation. Nature. 490, 49–54 (2012).
 Calcified shells provide critical protection against predation and desiccation in sessile marine animals such as oysters. Two models, matrix model and cellular model, have been advanced for molluscan shell formation. With "Proteome Profiling" the oyster shell proteome was completed, and 259 shell proteins were identified and mapped to the oyster genome. The role of cells in shell formation is further supported by the functional diversity of proteins detected in shells. Many shell proteins are enzymes that may be involved in matrix construction or modification. The results indicate that the oyster shell matrix is not formed simply by self-assembling silk-like proteins but by diverse proteins through complex assembly and modification processes that may involve haemocytes exosomes.
Calcified shells provide critical protection against predation and desiccation in sessile marine animals such as oysters. Two models, matrix model and cellular model, have been advanced for molluscan shell formation. With "Proteome Profiling" the oyster shell proteome was completed, and 259 shell proteins were identified and mapped to the oyster genome. The role of cells in shell formation is further supported by the functional diversity of proteins detected in shells. Many shell proteins are enzymes that may be involved in matrix construction or modification. The results indicate that the oyster shell matrix is not formed simply by self-assembling silk-like proteins but by diverse proteins through complex assembly and modification processes that may involve haemocytes exosomes.
Workflow:
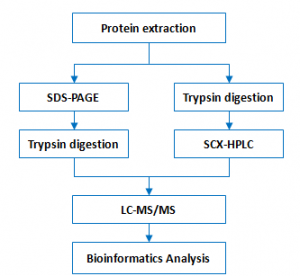
Bioinformatics:
Standard Bioinformatics Analysis
- Data statistics
- Protein identification by searching against database using MASCOT software
Advanced Bioinformatics Analysis
- Protein GO category analysis
- Protein COG category analysis
- Protein pathway analysis
Sample Requirements:
|
Sample Type |
Amount of Sample required |
Comment(s) |
|
Fresh animal tissue (wet weight) |
≥ 100 mg |
For tissue with high impurity or low amount of proteins such as plant root or phloem, 5g sample is needed. |
|
Fresh plant tissue or fungus (wet weight) |
≥ 1 g |
|
|
Microorganism (e.g. bacteria) |
≥ 200 mg |
|
|
Cell |
≥ 3-5×106 cells |
For phosphorylated protein identification, the cell number should be more than 107. |
|
Blood |
Serum / plasma ≥ 500 µl Whole blood ≥ 5 ml |
We do not recommend sending whole blood sample because blood cells can be broken during transport process. |
|
Please remove high-abundance protein before sending extracted proteins and attach gel images before and after the removal of high-abundance protein. |
||
|
Body fluid (e.g. saliva and cerebrospinal fluid) |
≥ 5 ml |
/ |
|
Urine |
≥ 50 ml |
Centrifuge at 1000g for 5min to discard the precipitate before sending your sample to BGI Tech. |
|
Protein extract |
≥ 500 µg (concentration ≥1mg/ml) |
/ |

